|
In general, neighboring NMR active nuclei three or fewer bonds away lead to this splitting. Nuclei which share the same chemical shift do not form splitting peaks in an NMR spectra. In NMR spectra, this effect is shown through peak splitting that can give direct information concerning the connectivity of atoms in a molecule. The easier way: subtract frequency 1 from frequency 3, and then 2 from 4. 
Different spin states interact through chemical bonds in a molecule to give rise to this coupling, which occurs when a nuclei being examined is disturbed or influenced by a nearby nuclear spin. Find the average of the two sets of outer lines, and take the difference. 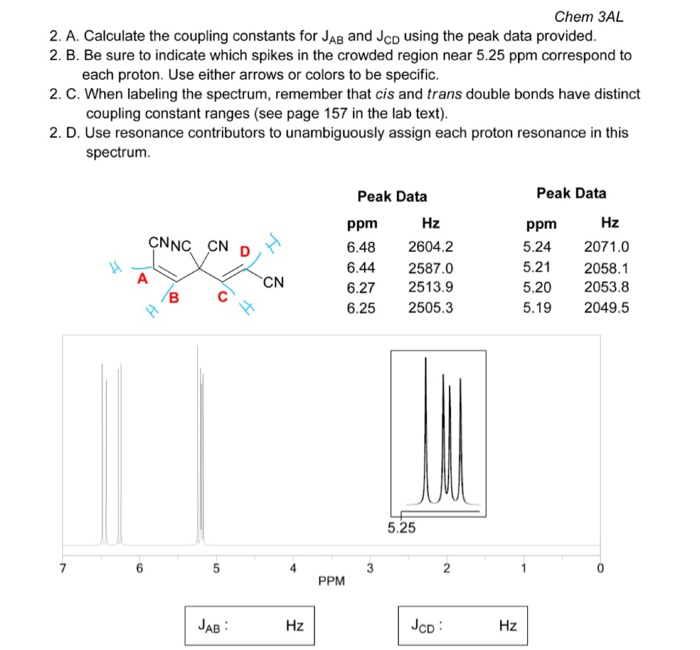
\): A 1H NMR spectra of ethanol (CH 3CH 2OH).Īnother useful property that allows NMR spectra to give structural information is called spin-spin coupling, which is caused by spin coupling between NMR active nuclei that are not chemically identical.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
